News
Members
Publications
Software / Data
Job offers
Images / Videos
Collaborations
Conferences
Lab meetings: "Les partages de midi"
Practical information
Members Area
Next conferences we are in …



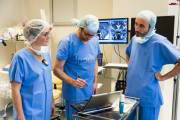
Survival prognosis from low grade glioma (LGG) is increased when the maximum tumoral tissue has been removed, slowing down evolution into high grade glioma (HGG), which is much more agressive (14 months mean survival rate). Intraoperative Positron Surface Imaging (PSI) has been suggested recently in neurosurgery to help the surgeon in detecting precise tumor margins. Originally designed in the mamography context by Pr. Dr. Nassir Navab (Munich, Germany), this method relies on scanning the resection cavity with a tracked freehand beta probe. In conjunction with a [18F]-based radiotracer, such system generates reconstructed surface images of the brain resection cavity. By introducing intraoperative freehand functional images in the operating room (OR), the resection gesture of the surgeon will be enhanced, driving to a better patient outcome.
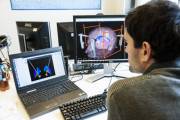
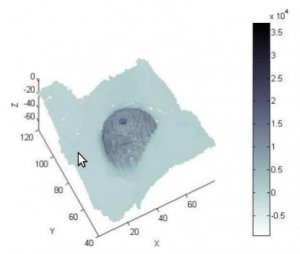
By comparison with existing intraoperative imaging modalities for LGG, PSI systems gives a fast solution to estimate resection marging, bringing functional information in the OR. PSI system provides reconstructed surface in real time (3 ms) after a 3 minutes scan of the surface of interest (SOI), but the image quality is less than satisfaying. In literature, radiation detection physics models have been introduce to improve the spatial resolution and the tumor contrast of reconstructions.
The main objective of our project is to perform the feasibility study of using PSI in the context of neurosurgery and especially the impact of detection model on image quality. A first step of this study concerns the evaluation of the technical parameter of the PSI system over over phantom configuration. sets of experiments have been used to: i) assessing and modelling the sensitivity of the beta probe and ii) studying the impact of reconstruction with different detection models on the probe response function. The technical evaluation outlines both optimal conditions of usage and contraints in the OR. On a second step, a validation study has been performed to enlight the detection model with the highest performances among existing models in literature. One of the previous studies provided a comparison between different acquisition models and enligthed the dramatic variation of reconstruction in freehand SPECT imaging. In this step, the performance has been detailled in term of efficiency, computation speed, spatial accuracy and tumor contrast, and evaluated in an intraoperative context over in-vitro data. Finally, the third step will evaluate performances of reconstructed images using realistic dataset. As a matter of fact, no clinical dataset has been acquired in the OR yet. To adress this issue, realistic resection brain cavity will be modeled and PSI acquisition will be simulated within the GEANT4 framework. Such study would act as pre-clinical validation before acquiring real data inside the OR.
In addition, our project aims to developp new acquisition models for PSI systems, dedicated to brain tumor surgery. From previous validation studies, reconstructions generated with detection models gave good performances in term of residual tumor detection, at the trade of high computation time (about 30 mn without GPU improvement). In the OR, the required computation time to generate images should be low to not interrup (that much) the surgical workflow. Thus, we proposed a new acquisition model more suitable for intraoperative usage. Our model gives lower computation time and better performances than the off the shelf original model.
 In this project, international collaborations and knowledge have been involved. On the one hand, CAMP team from the Technische Universitat Munchen gave us the opportunity to work on their PSI system and understand its behavior, while MediCIS team brings it skills in image guided neurosurgery. With this collaboration, acquisition on phantom and software engineering have been performed. On the second hand, a collaboration with a laboratory of the Harvard Medical Scool has been started. A validation framework has been developped in association with a neurosurgeon in Brigham and Women's Hospital in Boston, for future clinical validation, finalizing our feasibility study.
In this project, international collaborations and knowledge have been involved. On the one hand, CAMP team from the Technische Universitat Munchen gave us the opportunity to work on their PSI system and understand its behavior, while MediCIS team brings it skills in image guided neurosurgery. With this collaboration, acquisition on phantom and software engineering have been performed. On the second hand, a collaboration with a laboratory of the Harvard Medical Scool has been started. A validation framework has been developped in association with a neurosurgeon in Brigham and Women's Hospital in Boston, for future clinical validation, finalizing our feasibility study.


